In order to be able to determine the correct composition of the fertilizer medium, we need to know the optimal composition of the medium for a given phase of plant growth and growing conditions, as well as water quality parameters. It is necessary to know the salinity and pH of the water, as well as the concentration of bicarbonate (HCO3- ) and the content of macro and micronutrients. Bicarbonate data is needed to determine the amount of acid needed to acidify the nutrient solution.
In practice, it can be assumed to leave 43 mg/l (0.7 mmol/l) of free bicarbonate ions in the solution the remaining amount (depending on the content in the water) should be neutralized with the appropriate acid: nitric, phosphoric, or sulfuric acid. The amount of the component introduced with the acid should be taken into account in further calculations of the nutrient solution.
The amount of nitric or phosphoric acid needed to acidify water can be determined using the Acidification Calculator app located in the Play Store
| Potential problem | Units | Use without restrictions | Light and medium use | Heavy use |
|---|---|---|---|---|
| Salinity (EC) | mS/cm | < 0,7 | 0,7 – 3 | > 3,0 |
| Amount of dissolved salts | mg/l | < 450 | 450 - 2000 | > 2000 |
| Phytotoxicity to roots: | ||||
| sodium (Na) | SAR* | < 3 | 3 - 9 | > 9 |
| chlorine (Cl) | mg/l | < 140 | 140 - 350 | > 350 |
| boron (B) | mg/l | < 0,7 | 0,7 - 3,0 | > 3,0 |
| phytotoxicity to leaves: | ||||
| sodium (Na) | mg/l | < 70 | > 70 | - |
| chlorine (Cl) | mg/l | < 100 | > 100 | - |
* SAR (sodium adsorption ratio) determines the ratio of sodium to calcium and magnesium.
It is determined by the formula:
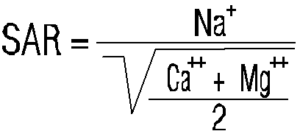
Water analysis should be carried out in a specialized chemical laboratory. Such services are provided min. Chemical Analysis Laboratory of the Institute of Horticulture - PIB in Skierniewice http://www.nawadnianie.inhort.pl/add/pdf/analizy_laboratoryjne.pdf laboratories of District Chemical and Agricultural Stations https://www.schr.gov.pl/index.php?c=page&id=31 and laboratories of private companies.